The electrons passing through the ETC gradually lose potential energy, and are finally deposited on the terminal electron acceptor, which is removed as waste from the cell. This process contributes to the creation of a transmembrane electrochemical gradient. These reactions couple the exergonic redox transfers to the endergonic transport of hydrogen ions across the membrane. The electrons are passed from enzyme to enzyme through a series of redox reactions. In aerobically respiring eukaryotic cells the ETC is composed of four large, multiprotein complexes embedded in the inner mitochondrial membrane and two small diffusible electron carriers shuttling electrons between them. ETCs are therefore the portion of respiration that use an electron acceptor (brought in from outside of the cell) as the final/terminal acceptor for the electrons that were removed from the intermediate compounds in catabolism. These "chains" of redox enzymes and electron carriers are called electron transport chains (ETC). In respiration, a special set of enzymes carry out a linked series of redox reactions that ultimately transfer electrons to the terminal electron acceptor. These redox reactions harvest energy for the cell by coupling exergonic redox reactions to an energy-requiring reactions in the cell. In between the original electron source and the terminal electron acceptor are a series of biochemical reactions involving at least one redox reaction (during the course of glycolysis, pyruvate oxidation, and the TCA cycle we saw many!). But once again, keep in mind that many organisms can use non-carbon based sources of electrons, and transfer the electrons to other kinds of oxidizing agents. We will often refer to these molecules as "fuel", and the analogy with the burning of gasoline, propane, or coal is quite apt these are all simply sources of electrons, and the waste product is CO 2 and water. 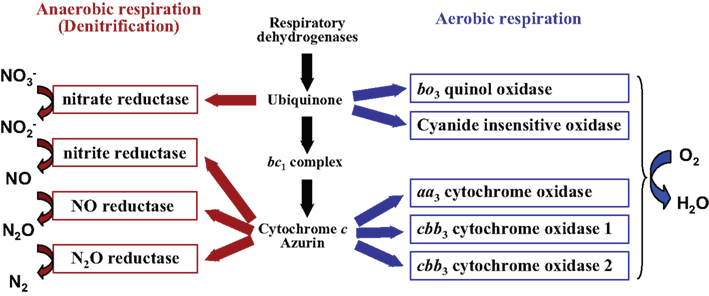
The molecules from which the electrons originate can vary greatly across biology (we have looked at one common source - the reduced carbon-based molecule glucose). These compounds where the electron is finally "dumped" are more generally known as " terminal electron acceptors". This generic idea can be generally applied across biology and oxygen need not always be the compound that brought in, reduced, and dumped as waste. Simply put, some reactant comes into the organism and then gets reduced and leaves the body as a waste product. When we breath, air-including molecular oxygen- is brought into our lungs from outside of the body, the oxygen then becomes reduced, and waste products, including the reduced oxygen in the form of water, are exhaled. A definition of the word "respiration" that most people are familiar with is "the act of breathing". In the next few modules we start to learn about the process of respiration and the roles that electron transport chains play in this process. A generic example of a simple, 2 complex ETC.The Electron and Proton carriers in the ETC.How do ETC complexes transfer electrons?.Using the redox tower: A tool to help understand electron transport chains.A quick summary of Electron Transport Chains.Introduction to redox, oxidative phosphorylation and Electron Transport Chains.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
